|
When an electric discharge is sent through a highly exhausted tube, the sides of the tube glow with a vivid green phosphorescence. The first place in which corpuscles were detected was a highly exhausted tube through which an electric discharge was passing. In this lecture I wish to give an account of some investigations which have led to the conclusion that the carriers of negative electricity are bodies, which I have called corpuscles, having a mass very much smaller than that of the atom of any known element, and are of the same character from whatever source the negative electricity may be derived. Nobel Lecture, Decemin Nobel Lectures: Physics, 1901-1921 (Amsterdam: Elsevier, 1967), pp. (See Pais 1986.) But Thomson did carry out this measurement and (later) the measurement of the particles's charge, and he recognized its importance as a constituent of ordinary matter. Thomson was not the only physicist to measure the charge-to-mass ratio of cathode rays in 1897, nor the first to announce his results. The credited discoverer makes crucial contributions to be sure, but often after fundamental observations have been made and tools invented by others. In what sense, then, can Thomson be said to have discovered the electron? After all, he did not invent the vacuum tube or discover cathode rays. Clearly, the characterization of cathode rays was a process begun long before Thomson's work, and several scientists made important contributions. The case of the electron raises several interesting points about the discovery process. He was awarded the Nobel Prize for physics in 1906 for this work, and in 1908 he was knighted. In 1899, he measured the charge of the particles, and speculated on how they were assembled into atoms. In 1897 he reported that "cathode rays" were actually negatively charged particles in motion he argued that the charged particles weighed much less than the lightest atom and were in fact constituents of atoms. For much of his career, Thomson worked on various aspects of the conduction of electricity through gases. Thomson was the Cavendish professor of Experimental Physics at Cambridge University and director of its Cavendish Laboratory from 1884 until 1919. 
Thomson, 1856-1940 see photo at American Institute of Physics) is widely recognized as the discoverer of the electron. The model has also been compared to a watermelon because the red edible part of a watermelon was compared to the sphere having a positive charge and the black seeds filling the watermelon looked similar to the electrons inside the sphere.Discovery of the Electron: J. It resembles a plum pudding because the electrons in the model look like the dry fruits embedded in a sphere of positive charge just like a spherical plum pudding. 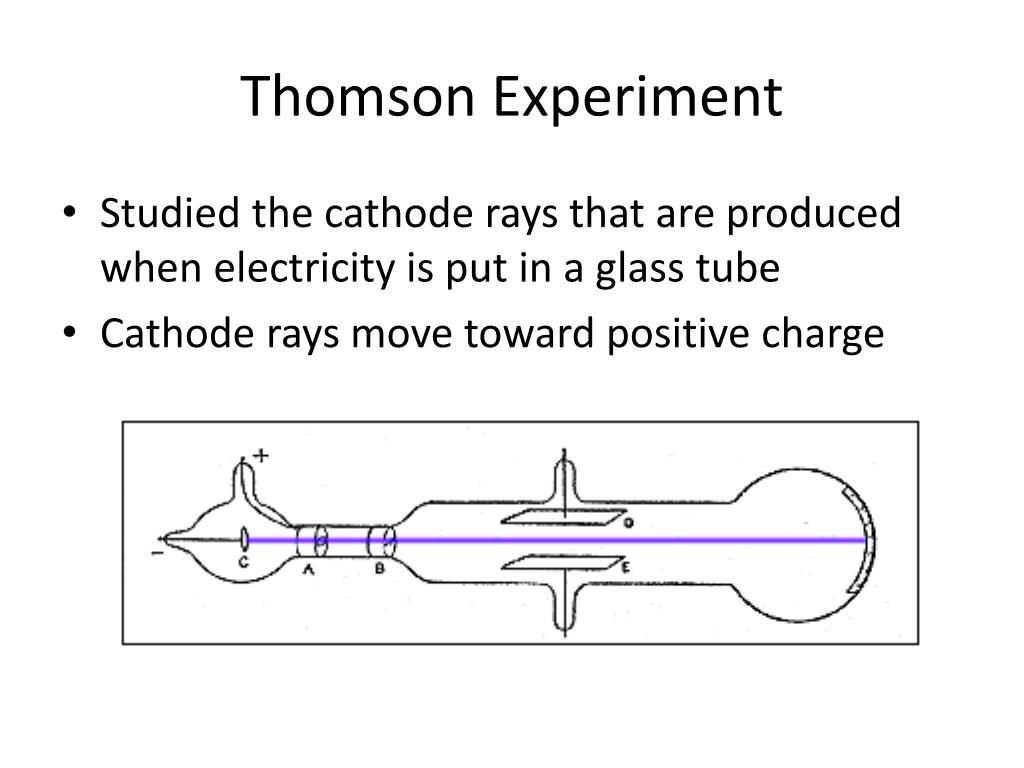
Thomson’s atomic model resembles a spherical plum pudding as well as a watermelon.The positive and negative charge is equal in magnitude and therefore an atom has no charge as a whole and is electrically neutral.According to the postulates of Thomson’s atomic model, an atom resembles a sphere of positive charge with electrons (negatively charged particles) present inside the sphere.You can download Structure of Atom Cheat Sheet by clicking on the download button below Learn about Charged particles in Matter in more detail here. Negatively charged particles called electrons are present in an atom.So, he proposed a model on the basis of known properties available at that time. However, at that time the atomic nucleus was yet to be discovered. It was proposed by J.J Thomson in the year 1904 just after the discovery of electrons. The description of Thomson’s atomic model is one of the many scientific models of the atom. Learn about the concept of an Atomic number here in detail. Towards Quantum Mechanical Model of Atom.Development Leading to Bohr’s Model of Atom.How are Electrons Distributed in Different Orbits (Shells)?. 
Browse more Topics under Structure Of Atom J.J Thomson and Rutherford first demonstrated the ionization of air in x rays. He assumed that an atom is composed of a cloud of negative charge in a sphere of positive charges. J.J Thomson believed electrons to be two thousand times lighter than a proton. Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. Thomson was the first and one of the many scientists who proposed models for the structure of an atom. The discovery of subatomic particles led to the search how the subatomic particles are arranged in an atom. However, the discovery of subatomic particles disapproved the postulates proposed in Dalton Atomic Theory. It explained atoms cannot be broken down into further smaller particles. Before the discovery of subatomic particles, John Dalton came up with Dalton’s atomic theory where he suggested that atoms are indivisible particles.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
